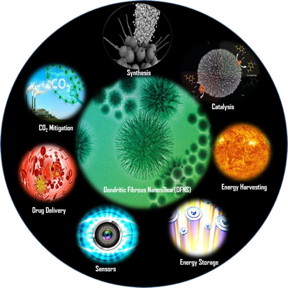
Dendritic fibrous nanosilica (DFNS) attracted a great deal of attention in a large number of scientific disciplines such as catalysis, solar energy harvesting (photocatalysis, solar cells, etc.), energy storage, self-cleaning antireflective coatings, surface plasmon resonance (SPR)-based ultra-sensitive sensors, CO2 capture, and biomedical applications (drug delivery, protein and gene delivery, bioimaging, photothermal ablation, Ayurvedic and radiotherapeutics drug delivery,
etc.). As discussed in this review, the unique fibrous morphology of the DFNS family of materials bestows them with several important properties that were brilliantly exploited for use in a range of applications. The fibers of DFNS were functionalized with a range of organic groups, ionic liquids, organometallic complexes, polymers, peptides, enzymes, DNA, genes, etc.
etc.). As discussed in this review, the unique fibrous morphology of the DFNS family of materials bestows them with several important properties that were brilliantly exploited for use in a range of applications. The fibers of DFNS were functionalized with a range of organic groups, ionic liquids, organometallic complexes, polymers, peptides, enzymes, DNA, genes, etc.
They were also loaded with metal nanoparticles, bi-metallic nanoparticles, even with single atoms of metals, quantum dots, and metal oxides and hydroxides. They were also used as hard templates for the synthesis of high surface area carbon with fibrous morphology. DFNS-based zeolites were also synthesized with unique
activities.
activities.
